Congress Newsletter 2023
EU-HyProtect heralds encouraging new data on predictive haemodynamic monitoring
Close intraoperative monitoring of arterial pressure is a clinical priority, since profound hypotension during surgery raises the risk of multiple negative sequelae including myocardial injury, acute kidney injury, and death.1 A significant recent advance in this area has been the development of predictive haemodynamic monitoring software, which aims to predict hypotension such that it can be averted even before it occurs. Encouraging new evidence regarding this software has recently been published in the form of data from the EU-HyProtect Registry.2
The EU-HyProtect Registry was set up to investigate the incidence, duration and severity of intraoperative hypotension in patients who were monitored with Acumen Hypotension Prediction Index software (Acumen HPI software; Edwards Lifesciences, Irvine, CA, USA) during elective major non-cardiac surgery. Acumen HPI software, which was developed using machine learning, predicts hypotension from features of the arterial pressure waveform. The likelihood of a patient developing hypotension (defined as a mean arterial pressure [MAP] of <65 mmHg for one minute or more) is rated on a unitless scale from 0 to 100, and if the value is over 85, acoustic and visual alarms are triggered. To guide rapid treatment, a pop-up window also displays additional haemodynamic variables which can help clinicians determine the cause of hypotension, and react accordingly.
Karim Kouz, first author of the recent publication on EU-HyProtect study,2 explains how predictive monitoring with this software compares to traditional reactive monitoring. “Previously, we would measure blood pressure, and when we detected hypotension, we would give treatment – but by then some harm would have already occurred to the patient,” he says. “The advantage of this predictive software is that we can treat upcoming hypotension without the patient actually becoming hypotensive.”
To examine the software’s performance in the clinic, Dr Kouz and colleagues built the EU-HyProtect Registry. In this prospective observational study, a total of 702 patients were included from 12 medical centres in five European countries (Figure 1).
All patients underwent elective major non-cardiac surgery, predicted to last at least 120 minutes, under general anaesthesia requiring continuous invasive blood pressure monitoring. The primary endpoint was the time-weighted average MAP below a threshold of 65 mmHg, calculated as the area of the curve, divided by the total duration of surgery. This measure was chosen as it encapsulates both the duration and severity of hypotension, with a correction for the total monitoring time.
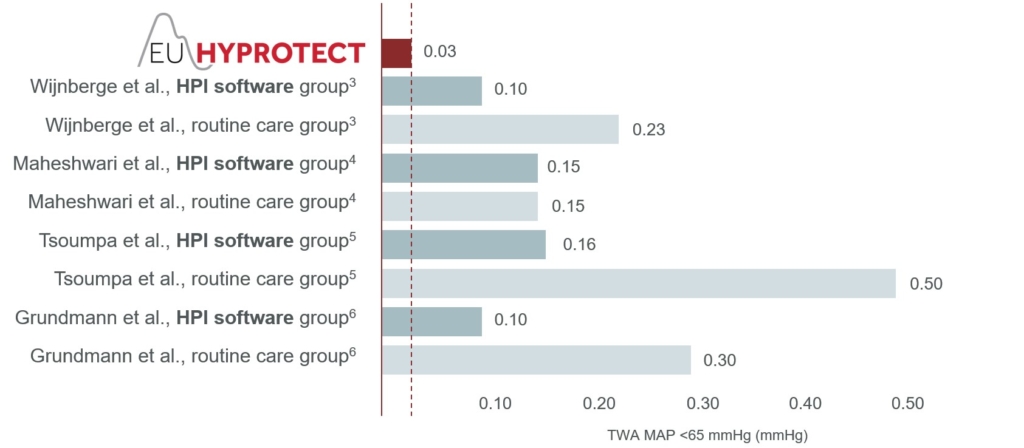
The headline finding of the study was that the median time-weighted average MAP <65 mmHg was remarkably low: just 0.03 mmHg (interquartile range [IQR] 0.00–0.20 mmHg; Figure 2). Additional analyses revealed that patients spent a median of only 2.0 min (IQR 0.0–9.1 min) or 1% (IQR 0–5%) of surgical time below the threshold MAP of 65 mmHg. Additionally, 41% of patients experienced no episodes of at least one minute with MAP <65 mmHg, and the median number of such episodes was 1 (IQR 0–3). The median absolute maximum decrease in MAP below 65 mmHg lasting ≥1 minute was 7 mmHg (IQR 3–12).2
Dr Kouz highlights the implications of these results. “Overall, we found that the duration and severity of hypotension were very low when the Acumen HPI software was used in a real-world setting,” he says. “Given our data, we can be highly hopeful that Acumen HPI software could help to reduce the duration and severity of hypotension in these patients.”
To place these findings in context, in three randomised trials evaluating Acumen HPI software for intraoperative monitoring,3–5 two found that use of the software reduced intraoperative hypotension.3,5 All three studies reported a notably higher median time-weighted average MAP <65 mmHg than was found in the EU-HyProtect study. Values of 0.443, 0.144 and 0.505 mmHg were reported in patients assigned to the routine care arms of these three trials, and 0.103, 0.144 and 0.165 mmHg in those patients having monitoring with Acumen HPI software.
Considering these results, it is interesting that the values in the intervention arms as well as the control arms of these three trials were substantially higher than the 0.03 mmHg found in the EU-HyProtect study. A potential reason for the remarkably low burden of hypotension in EU-HyProtect could be the beneficial effect of familiarity with the software, as the registry only recruited patients in medical centres where Acumen HPI software was routinely used for haemodynamic monitoring. Whether or not this fully explains the difference, it is encouraging that this observational registry study found the real-world duration and severity of intraoperative hypotension to be very low with Acumen HPI software.
With regards to adding to the research landscape, the fact that the EU-HyProtect study collected real-world evidence is a key benefit, as is its large sample size. The three previously published randomised trials mentioned above recruited between 68 and 214 patients, while a previous retrospective observational study had a sample size of 100,6 so this registry data from over 700 patients represents a valuable addition to our understanding of predictive haemodynamic monitoring.
The principal investigator of EU-HyProtect, Bernd Saugel, comments on the unique features of this study and the implications for further research. “This was the first observational registry study of Acumen HPI software, including several hundred patients across multiple European countries,” he says. “The encouraging results indicate that we should go on to perform large randomised trials with this software.” This will enable to compare hypotension between patients monitored with Acumen HPI software and those in routine care, to confirm whether there is a causal relationship between predictive monitoring and hypotension reduction. Crucially, future trials can also examine whether effects on arterial pressure translate into improved patient outcomes in terms of reduced morbidity and mortality.
References
- Gregory A, Stapelfeldt WH, Khanna AK, et al. Anesth Analg 2020;132:1654–65.
- Kouz K, Monge García MI, Cerutti E, et al. BJA Open 2023;6:100140.
- Wijnberge M, Geerts BF, Hol L, et al. JAMA 2020;323:1052–60.
- Maheshwari K, Shimada T, Yang D, et al. Anesthesiology 2020;133:1214–22.
- Tsoumpa M, Kyttari A, Matiatou S, et al. J Clin Med 2021;10:58.
- Grundmann CD, Wischermann JM, Fassbender P, et al. Acta Anaesthesiol Scand 2021;65:1404–12.
For more information, please visit https://www.edwards.com/gb/devices/hemodynamic-monitoring.
PP–EU-6414 v1.0